2025 Highlights
Montefiore Einstein Obstetrics & Gynecology and Women’s Health
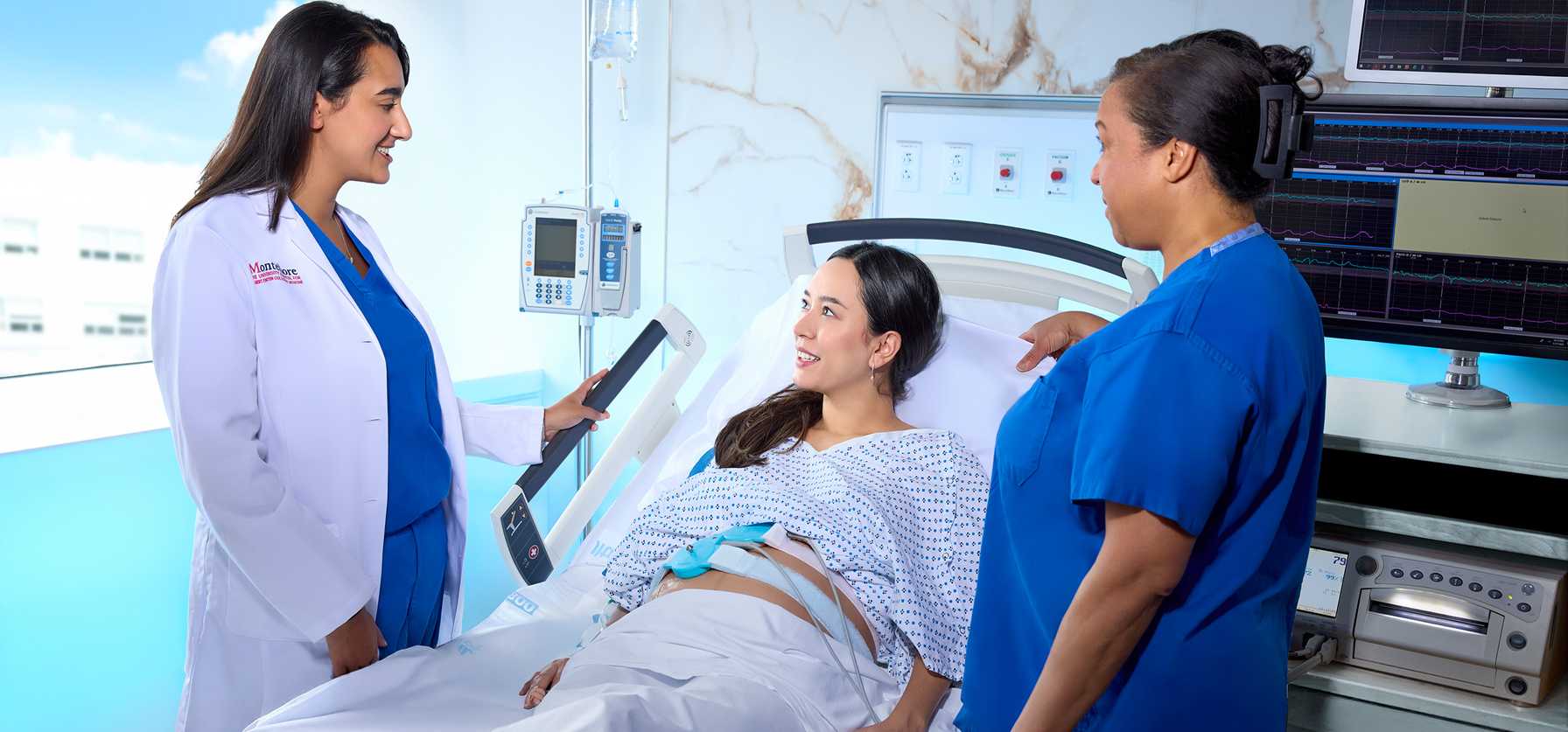
Setting the Standard in Expertise – For Every Stage of a Woman's Life
We’re ranked in the top 1% of all hospitals in the nation for obstetrics and gynecology and among the highest performing hospitals in the nation for maternity care, according to U.S. News & World Report. Our Gynecologic Oncology Program is fully integrated with our Montefiore Einstein Comprehensive Cancer Center, among the elite 1% National Cancer Institute (NCI)-designated comprehensive cancer centers in the U.S.
Mark Einstein, MD, MS, Chair, Department of Obstetrics & Gynecology & Women's Health
Note from the Chair
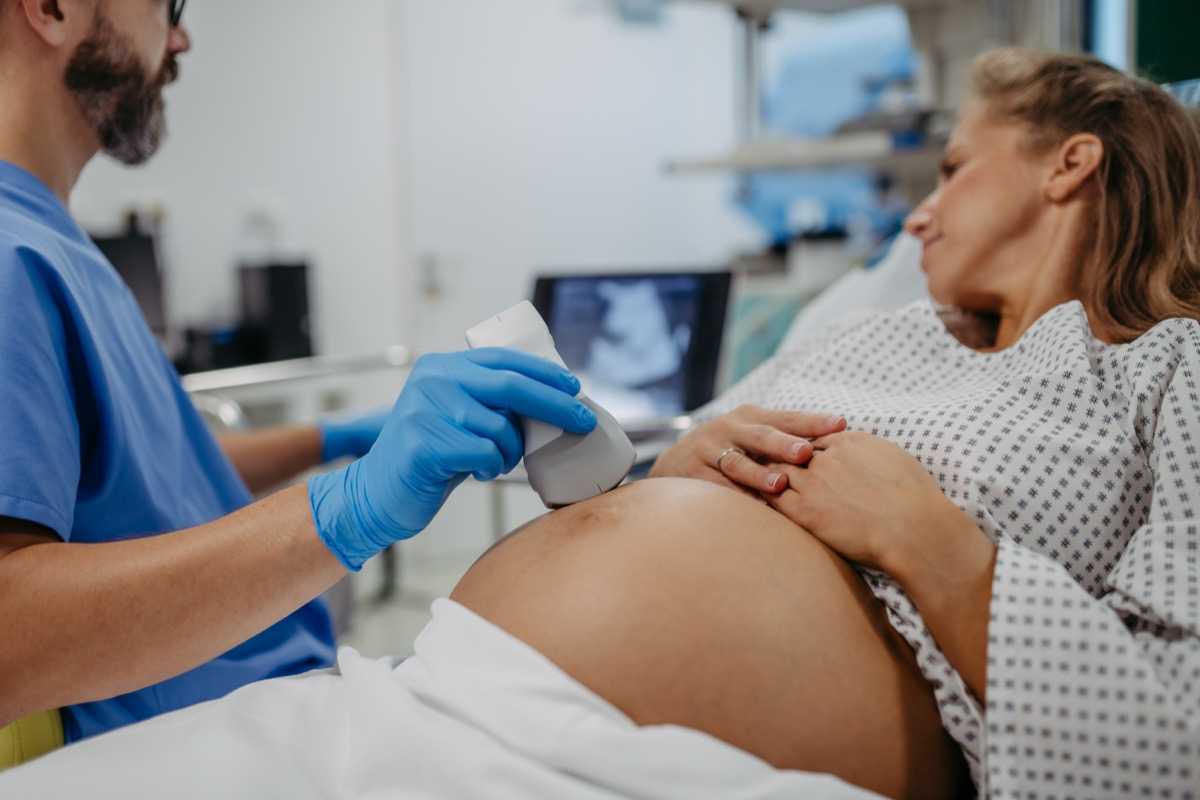
In 2025, Montefiore Einstein’s Department of Obstetrics & Gynecology & Women’s Health expanded multidisciplinary high-risk services, scaled minimally invasive surgical capacity, and continued robust clinical and translational research — while maintaining a sustained commitment to equitable, patient-centered care across the Bronx, Westchester, and the Hudson Valley.
DATA & STATISTICS
By the numbers
3,334
Delivery volume at our Weiler & Wakefield Campuses
924
Minimally invasive and robotic procedures
7
Peer-reviewed publications between July 1,2024 – June 30, 2025
3
Clinical trials
Leadership
Division Directors
Education Program Directors
Administration
Innovations and achievements
Pioneering the future of treatment and care
Advancing care through clinical trials
We are spearheading all phases and types of clinical trials, including several sponsored by the NIH, translating novel scientific discoveries into new diagnostics and treatments to help improve clinical outcomes. By enrolling in a clinical trial, you can help our scientists develop new treatments, diagnostic tools and other strategies to prevent, diagnose and treat maternal, fetal, obstetric, gynecologic and women’s health conditions. Through our clinical trials, you may also be able to access emerging technologies and innovative treatments that may otherwise not be available to the general public.
Highlights from our current active clinical trials include:
|
NCI Cervical Cancer Last Mile Initiative Self-collection for HPV testing to Improve Cervical Cancer Prevention (SHIP) Trial - Sub-study S03
|
Performance of Simultaneous Saline-Infusion Sonography and Endometrial Sampling (SISS) for the evaluation of Endometrial Pathology in patients with post menopausal bleeding (Investigator Initiated Study)
|
|
Post Cesarean Delivery Uterine deformation and risk of malposition of Intra Uterine Device IUD
|
Claudin-6 Overexpression in Endometrial and Ovarian Cancers and Associated Prognostic Value A Single Institution Study of a High-Risk Diverse Population
|
|
The significance of a pathology report of proliferative endometrium and insufficient endometrial tissue in the endometrial sampling of women with post-menopausal bleeding PMB
|
Patient Opinions Regarding Genetic Services for Hereditary Ovarian Cancer Risk in a Racially, Ethnically and Economically Diverse Population
|
|
The performance of endometrial sampling and radiographic evaluation in uterine carcinomas and other uterine cancer
|
First trimester screening for Morbidly Adherent Placenta (MAP)
|
|
Screening performance of first trimester ultrasonography in the diagnosis of Morbidly Adherent Placenta (MAP)
|
Patient acceptability and end-of-life outcomes after administration of an online goals-of-care module compared to traditional palliative care counseling
|
|
Understanding Perspectives and Experiences of Patients with Peripartum Cardiomyopathy
|
Phase I/Ib Trial Evaluating the Safety and Efficacy of BET Inhibitor, ZEN003694 With PD-1 Inhibitor, Nivolumab With or Without CTLA-4 Inhibitor, Ipilimumab in Solid Tumors
|
|
Effect of Validated Skills Simulation with the Miya Model on Operating Room Performance of Vaginal Hysterectomy
|
Addressing Fertility Preservation Concerns and Barriers of Cancer Patients in our Catchment Area
|
|
HyDROPS (Hydrops Diagnosing and Redefining Outcomes with Precision Study)
|
Human papillomavirus (HPV) transcriptional biomarkers for predicting the development of cervical dysplasia
|
|
A randomized controlled trial of bupivacaine with epinephrine for local pain control following perineal laceration repair in patients with pre-existing epidural analgesia (PAIN Trial)
|
Preoperative Mindfulness Training for Minimally Invasive Hysterectomy: A Randomized Controlled Trial
|
|
A Single Arm, Open-Label Pivotal Contraceptive Efficacy Study of Ovaprene
|
Mifepristone Versus Misoprostol for Cervical Preparation Prior to Procedural Abortion at 12 to 16 weeks Gestation in an Academic Medical Center
|
|
An Open-Label Study of E7386 in Combination With Other Anticancer Drug(s) in Subjects With Solid Tumors
|
ACR-2316-101 Phase 1 study of ACR-2316 in subjects with advanced solid tumors
|
|
Enhancing Opportunities to Identify Lynch Syndrome in a Racially Diverse Population through Endometrial Hyperplasia Tissue Molecular Testing
|
The Heart Outcomes in Pregnancy Expectations (H.O.P.E) for Mom and Baby Study
|
|
Hereditary Cancer Screening in an Urban Cancer Survivorship Clinic A Quality Improvement Project
|
The role of HPV typing and cytology in anal cancer screening in individuals living with HIV
|
|
NRG-GY018 - Testing the Addition of the Immunotherapy Drug Pembrolizumab to the Usual Chemotherapy Treatment (Paclitaxel and Carboplatin) in Stage III-IV or Recurrent Endometrial Cancer
|
NRG-GY028 - A Phase IB and Randomized Phase II Trial of Megestrol Acetate with or Without Ipatasertib in Recurrent or Metastatic Endometrioid Endometrial Cancer
|
|
A Study of CBX 12 in Subjects With Platinum Resistant or Refractory Ovarian Cancer
|
A Study of E7386 in Combination With Other Anticancer Drug(s) in Participants With Solid Tumor
|
|
GOG-303 - A Study to Evaluate Dostarlimab Plus Carboplatin-paclitaxel Versus Placebo Plus Carboplatin-paclitaxel in Participants With Recurrent or Primary Advanced Endometrial Cancer (RUBY)
|
GOG3053 - Study of Pembrolizumab (MK-3475) in Combination With Adjuvant Chemotherapy With or Without Radiotherapy in Participants With Newly Diagnosed Endometrial Cancer After Surgery With Curative Intent (MK-3475-B21 / KEYNOTE-B21 ENGOT-en11 / GOG-3053)
|
Research Review
Improving care and outcomes through research
In 2025, we published a multitude of scientific articles across a variety of topics.
Explore our latest
research.
Patient referrals
At Montefiore Einstein Obstetrics & Gynecology and Women’s Health, we know that providing patients with the best possible care includes teamwork and trust. We work closely with our valued referring physicians to ensure open communication and reliable expertise.
Contact us
Linda Gillespie, MSEd, MBA
Director, Patient Experience, Access and Special Projects, Obstetrics, Gynecology and Women’s Health, Montefiore Einstein
LGILLESP@montefiore.org